Proprio - Paradigm
Overview
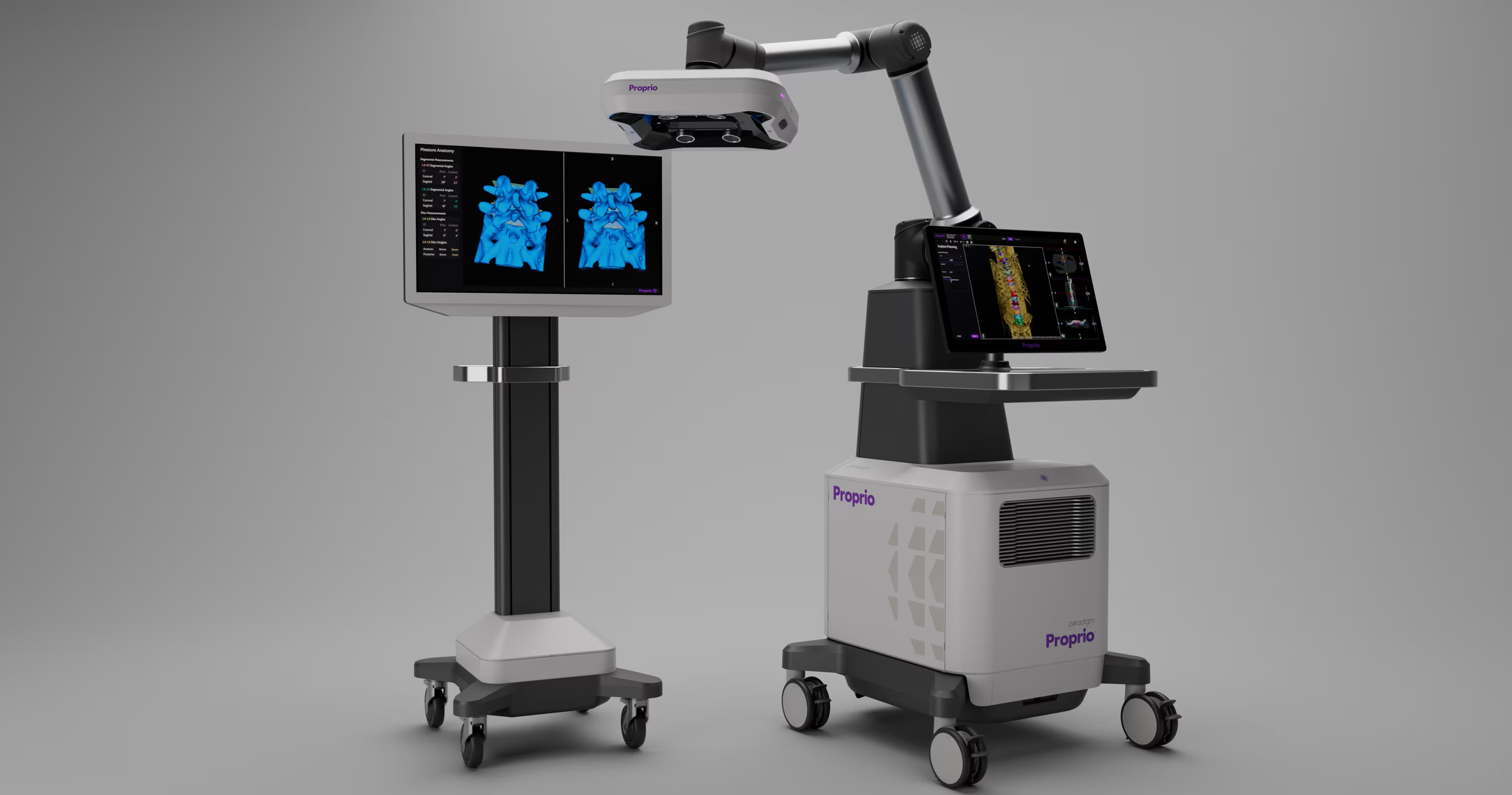
At Proprio, I worked on the Paradigm system, a computer-vision–based surgical platform designed to improve intraoperative decision-making while reducing reliance on radiation-based imaging. The system fused infrared instrument tracking, RGB imaging, and depth-based spatial reconstruction to generate a real-time 3D model of the surgical field.
Within this reconstructed environment, surgeons could obtain real-time alignment measurements and navigate instrumentation directly in 3D space without requiring repeated radiation during the procedure. By maintaining continuous spatial awareness through tracked instruments and sensor fusion, the system reduced the need for repeated intraoperative imaging to verify positioning and alignment.
My Role & Ownership
I served as the architectural owner of the user-facing application layer for Paradigm. My responsibility was translating complex, sensor-derived spatial data into stable, reliable, and clinically usable workflows.
I worked closely with the design team to evaluate feasibility, shape interaction constraints, and align implementation timelines with clinical and regulatory requirements. This required balancing real-time performance considerations, system accuracy, and maintainability while ensuring the resulting experience remained intuitive for surgeons.
Regulatory & Stability
Paradigm operated within a regulated medical device environment, and the intraoperative application was part of multiple FDA 510(k) submissions. During my time on the product, I contributed to four 510(k) submissions and numerous Letters to File.
I owned software risk recognition and mitigation for the intraoperative user-facing application. This involved identifying failure modes specific to real-time visualization and navigation, defining appropriate mitigations, and ensuring those mitigations were implemented and verifiable.
While the design team authored product requirements, I was responsible for validating that proposed requirements were technically sound, implementable within system constraints, and appropriately traceable to software artifacts. I maintained traceability from high-level requirements through implementation and testing, ensuring alignment between clinical intent, regulatory documentation, and delivered behavior.
In practice, this meant balancing forward feature development with stability hardening, documentation rigor, and controlled release processes required in a regulated environment.
Case Studies
-
FERA
Led the incremental rearchitecture of a live medical device frontend to improve testability, stability, and long-term maintainability without disrupting clinical use.
-
Picasso
Introduced hands-free surgical control through a scalable, hardware-abstracted input system that integrated cleanly with the application’s MVVM architecture.